(Elektronika Dasar)
Electricity
Electricity
Back to Basics
Electricity is an essential ingredient of matter. The best way to understand the nature of electricity is to examine the smallest component of every element, the atom.
This is a Lithium atom.The third simplest atom after Hydrogen and Helium, Lithium atoms have 3 electrons that encircle a nucleus of 3 protons and 4 neutrons.
- Electrons have a negative electrical charge.
- Protons have a positive electrical charge.
- Neutrons have no electrical charge.
Ions
Normally an atom has an equal number of electrons and protons. The charges cancle to give the atom no net electrical charge. It's possible to dislodge one or more electrons from most atoms. This causes the atom to have a net positive charge. It's then called a positive ion.
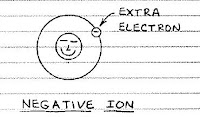
If a stray electron combines with a normal atom, the atom has a net negative charge and is called a negative ion.
Electrons
Free electrons can move at high speed through metals, gases and a vacuum, or they can rest on a surface.
More about free electrons
Many trillions of electrons can rest on a surface or travel through space or metter at near the speed of light (186,000 miles per second)
Resting Electrons
A group of negative electrons on a surface causes the surface to be negatively charged. Since the electrons are not moving, the surface can be said to have a negative static electrical charge.
Moving Electrons
A stream of moving electrons is called an electrical current. Resting electrons can quickly form an electrical current if placed near a cluster of positive ions. The positively charged ions will attract the electrons which will rush into fill the "holes" or void left by the missing electrons.
Missing Electrons
Mechanical friction, light, heat or a chemical reaction may remove electrons from a surface. This cause the surface to be positively charged. Since the positively charged atoms are at rest, the surface can be said to have a positive static electrical charged.
to be continued.. Simple Basic Electronics (part 2)






No comments:
Post a Comment